 
9.5.2.4.4 ReclaimerĪ side stream of from 1% to 3% of the MEA circulation is drawn from the bottom of the stripping column. A reclaimer is often included to remove these contaminants. Normal regeneration temperature in the still will not regenerate heat-stable salts or oxazolidone-2. At temperatures above 245 ☏ (118 ☌) a side reaction with CO 2 exists that produces oxazolidone-2, a heat-stable salt, which consumes MEA from the process. MEA reacts with carbonyl sulfide (COS) and carbon disulfide (CS 2) to form heat-stable salts, which cannot be regenerated at normal stripping column temperatures. Acid gases evolve into the vapor and are removed from the still overhead. Reactions with CO 2 and H 2S are reversed in the stripping column by heating the rich MEA to ~ 245 ☏ at 10 psig (118 ☌ at 69 kPa). MEA reactions are reversible by changing the system temperature. MEA is a stable compound and, in the absence of other chemicals, suffers no degradation or decomposition at temperatures up to its normal boiling point. The process is well proven and can meet pipeline specifications. MEA is a primary amine, which has had widespread use as a gas sweetening agent. PhD, PE, in Surface Production Operations (Third Edition), Vol9.5.2.4 Monoethanolamine Systems 9.5.2.4.1 General Discussion Oxidation inhibitors, nitrogen sparging to remove dissolved oxygen, or other methods will be required to manage oxidation. Unfortunately, MEA is prone to oxidative degradation and its T max for thermal degradation is 120☌. Its heat of CO 2 absorption is high, so it performs well with thermal-swing regeneration. It is a primary amine near the optimum Acid Dissociation Constant (pKa), so it has an average rate of CO 2 absorption and above average normalized capacity with excellent viscosity. It is produced in large quantities from the reaction of ammonia and ethylene oxide, a primary raw material for antifreeze polyester. MEA is the least expensive of the important absorption liquids. Numerous publications report extensive bench-scale and pilot plant results with MEA. Fluor has commercialized 30 wt% MEA in a number of units based on gas combustion. ASEA Brown Boveri (ABB) Lummus built several small units scrubbing coal-fired flue gas with 20 wt% MEA using carbon steel with a corrosion inhibitor. MEA is an important candidate for CO 2 capture by amine scrubbing. Rochelle, in Absorption-Based Post-combustion Capture of Carbon Dioxide, 2016 3.9.1 Monoethanolamine (MEA) The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.Conventional amine scrubbing for CO2 capture If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Ī common request on this site is to convert grams to moles. These relative weights computed from the chemical equation are sometimes called equation weights. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. 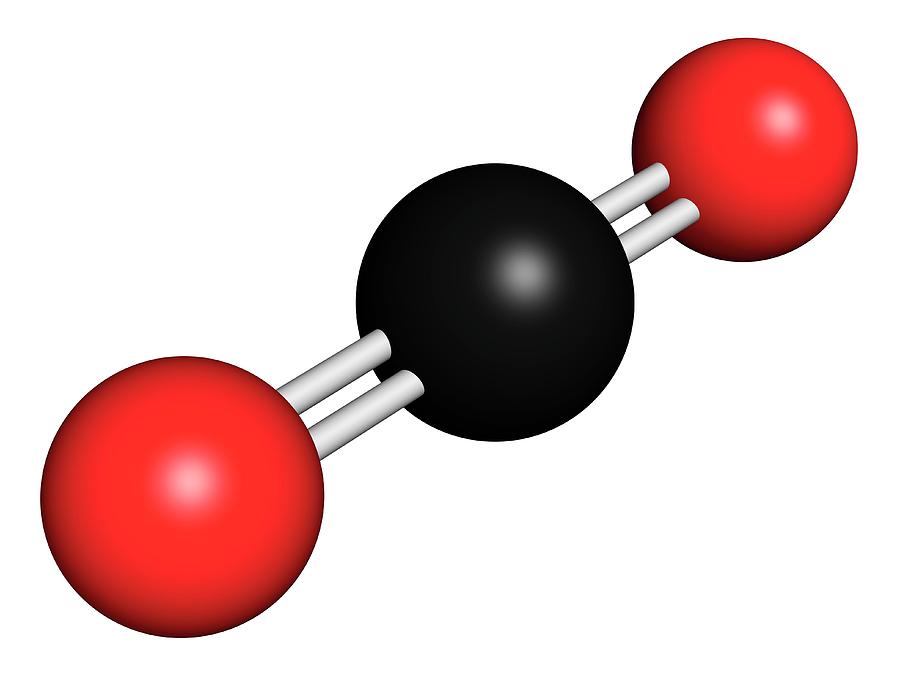
In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įinding molar mass starts with units of grams per mole (g/mol). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
